 
In particular, unsaturated vegetable oil can be interesterified with a fully hydrogenated version thereof, as in the illustration to the right. Typically the feedstock (starting product) is a mixture of two or more oils. Feedstock Īn example of interesterification between a triglyceride (top left) with two linolenic acid residues (LARs) and one stearic acid residue (SAR), and another triglyceride (bottom left) with three SARs, yielding two molecules with two SARs and one LAR each (right). Interesterified fats are used in many industrial food products, including cookies, crackers, biscuits, cakes and icings, dairy fat replacers, pie crust, popcorn, flatbread and tortillas. Also, some of the new triglycerides produced by IE may be fractionated (separated) through controlled crystallization. However, those other techniques may still be applied to the starting fats or to the products of IE, and the latter may be blended with other fats. In contrast to hydrogenation, interesterification is generally the original distribution of fatty acids in the product and hence is expected to preserve its nutritional and health attributes. It can also be used to prevent separation of solid fractions in palm oil and lauric fats, slow rancidification, or create oils more suitable for deep frying. It can be used, for instance, to turn oils into solid or semisolid products by combining them with other solid fats. This process is typically used to adjust the physical characteristics of the fat, such as melting point and plasticity, for specific uses. These reactions are performed by inorganic catalysts, yielding what is called chemical interesterification (CIE) in the industry or by enzymes, in the so-called enzymatic interesterification (EIE). The process implies breaking and reforming the ester bonds C–O–C that connect the fatty acid chains to the glycerol hubs of the fat molecules. In the food industry and biochemistry, interesterification (IE) is a process that rearranges the fatty acids of a fat product, typically a mixture of triglyceride. 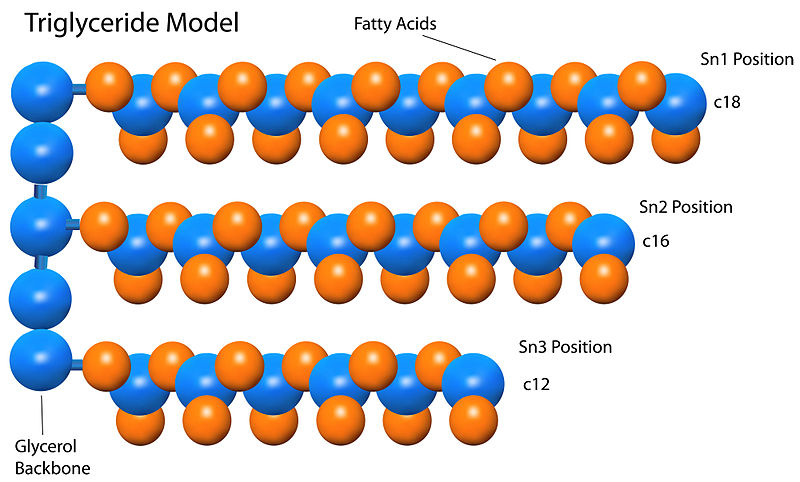
This article is about the chemical process. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
